- Joined
- 10 March 2009
- Posts
- 521
- Reactions
- 579
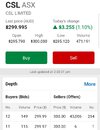
January update for the 2024 stock tipping comp. CSL has recovered slightly up 3.8% so far this month. On track to give a modest return over the next few months. On track from my perspective
Iggy
Iggy